INTENDED PURPOSE
Swab Intended purpose is “MEDICAL DEVICE” CLASS IIA STERILE. Swabs are sterile and ready-for-use systems intended for clinical samples drawing, transport and maintenance for cultural exam. The swab is suitable for short contact with the
human body including surgical wounds. Test Tubes are classified as IVD and are intended only for the preservation of samples. Product must be used as directed, Microbiotech is not responsible for any unauthorized use.
For professional use only.
National classification of medical devices (CND - For Italian law) > V901302 (Transport swabs for collection sample)
APPLICATOR
Special polyurethane foam, medical grade, swab tip that allows a better clinical sample collection and the following immediate release into the liquid transport medium. The soft foam bud is more comfortable for patients, and has significant
advantages for both conventional and molecular methods. It has a plastic shaft with "breakpoint" to allow the swab breaking into the test tube and the successive collection by the screw "capture" cap after the tube closing. It allows to dispose
of the swab together with the cap in a safer way.
Available in two versions on sampling site base:
- Standard tip: swabs, standard dimensions, for buccal, nasal, throat, vagina, rectal, cutaneous and wound sampling; this specialty applicator is tipped with medical grade polyurethane foam. It is bonded securely to the plastic handle. The best choice for buccal swab, for general specimen collection for testing and screening. Used in Pediatrics, Opthamology, Veterinary care, and oropharyngeal applications.

Overall Length: 152.4 mm (6”)
Tip Ø 4.775 mm (0.188”) - Length 16.002 mm (0.63”)
Handle Ø 2.54 mm (0.1”) - Length 149.225 mm (5.875”)
- Fine tip: swabs, small dimensions, for paediatric, urethra, urogenital and nasopharyngeal sampling. The special small applicator is tipped with medical grade polyurethane foam. It is bonded securely to the plastic handle. The best choice for paediatric, urethral and nasopharyngeal applications.

Overall Length 152.4 mm (6.”)
Tip Ø 1.981 mm (0.078”) - Length 17.45 mm (0.687”)
Handle Ø 2.159 mm (0.085”) tapered to Ø 1.016 mm (0.040”) - Length 148.539 mm (5.848”)
TEST TUBE
Liquid Transport Medium is contained in a cylindrical test tube Ø 13x80 mm, in transparent, unbreakable medical polypropylene (PP), to preserve sample and users safety. With screw cap in polyethylene (HDPE)
with “capture cap” technology, The specific color cap identifies instantly and secure the medium content. Pre-labelled on test tube to allow a correct patient and sample identification. Item code, lot number
and expiry date printed on each label for a full tracciability. 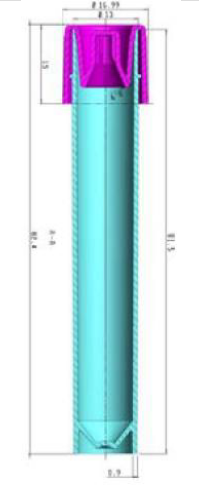
VIRUS LIQUID TRANSPORT MEDIUM
Swabs with red screw cap test tube containing 1 or 3 ml of liquid VIRAL transport medium for the collection and transport of clinical samples containing viruses,
chlamydiae, mycoplasmas and ureaplasmas from the collection site to the testing laboratory. The specimen transported in the Foam Virus Liquid Swabs can be used in the
laboratory to perform viral, chlamydial, mycoplasmal and ureaplasmal culture.
Viral transport medium, stable at room temperature, consists of a balanced buffer solution to maintain neutral pH, anti-microbial agents and a preservative.
Organisms viability maintained at room temperature and on ice. Suitable also for long term freeze storage. This product is not intended to be used for the collection and
transport of general bacterial and fungal specimens.
RESULTS
The survival and recovery of viruses, chlamydiae, mycoplasmas and ureaplasmas depends upon a series of factors such as infectious organisms type, transport time, preservation temperature, infectious organisms concentration in the sample.
Specimens should be collected soon after the onset of symptoms when microorganism titers are at their highest.
Specimens should be placed in the transport medium immediately following collection and promptly transferred to the laboratory for processing. For optimum recovery, specimens should be refrigerated during transport.
For long term storage, specimens should be frozen at -70°C or colder.
Use of the Foam Virus Liquid Swabs in conjunction with rapid diagnostic kits and instruments must be validated prior to use by the user.
PRECAUTIONAL MEASURES
- Transport medium (if present) and the defined sampling site;
- Use only for the collection and transport of human biological samples for the detection of potential pathogens such as viruses, chlamydiae, mycoplasmas and ureaplasmas, compatible with the type of device selected;
- Do not deviate from the intended use;
- Do not use the product if it is expired or the package is opened/damaged. Sterility guaranteed if unopened;
- Use the device following aseptic procedures;
- Single-use device; do not reuse. Reusing the device could contaminate the sample and/or the patient;
- Keep the device away from flames and heat sources;
- The fiber is only guaranteed to adhere to the shaft for instant sampling;
- The shaft is breakable, exert moderate pressure during sample collection;
- Store in a cool, dry place at a temperature between +2°C (35.6°F) and +25°C (77°F). Do not freeze;
- After use, the device may contain infectious microorganisms. Use appropriate PPE and dispose of the test tube and swab according to current regulations for medical waste.
- Keep out of flame or heat sources which might damage the product.
- Keep out of reach of children.
- Strictly follow the user’s instructions.